You have no items in your shopping cart.
Isocitrate Dehydrogenase (IDH): The Crucial Link Between Metabolism and Cancer
Technical Insight | Cellular Metabolism & Oncology
Isocitrate dehydrogenase (IDH) is a key regulatory enzyme of the tricarboxylic acid (TCA) cycle. It catalyzes the oxidative decarboxylation of isocitrate to α-ketoglutarate (α-KG), producing NADH (or NADPH) and CO2. Beyond its traditional role in energy production, IDH has emerged as a central player in cellular redox homeostasis and epigenetic regulation.
Structural Characteristics and Enzymatic Function
1. NAD+-dependent IDH
Located in the mitochondrial matrix, this isoform is a classic engine of the TCA cycle, fueling oxidative phosphorylation through NADH generation.
2. NADP+-dependent IDH
Found in both cytosol and mitochondria, it provides the NADPH necessary for reductive biosynthesis and protecting cells from oxidative stress.
The catalytic mechanism involves dehydrogenation followed by decarboxylation. This reaction is irreversible under physiological conditions, establishing IDH as a critical metabolic checkpoint.
Biological and Metabolic Significance in Oncology
IDH activity is tightly regulated by the cellular energy charge: ADP acts as an allosteric activator, while ATP serves as an inhibitor.
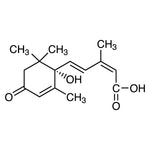
The Oncometabolite Connection: Mutations in IDH1 and IDH2 confer a neomorphic activity that converts α-KG into 2-hydroxyglutarate (2-HG). The accumulation of 2-HG disrupts epigenetic regulation, contributing to the development of gliomas and acute myeloid leukemia (AML).
Clinical and Industrial Relevance
-
•
Clinical Diagnostics Elevated serum IDH activity is a recognized biomarker for liver disease and specific malignancies.
-
•
Industrial Biotechnology In citric acid fermentation, managing IDH feedback inhibition is essential for maximizing production efficiency.